What are cannabinoids?
Cannabinoids are organic compounds of a lipid nature, capable of modulating a signaling system in the human body: the endocannabinoid system (ECS). This system participates in the regulation of processes such as neurotransmission, immune response and inflammation, energy metabolism, nociception (pain perception), and stress response.
The mechanism of action of cannabinoids is based on their ability to bind to specific receptors distributed throughout different tissues. This interaction can activate or inhibit intracellular signaling cascades, altering neurotransmitter release, gene expression, or the inflammatory response. The specificity of the effect depends on the type of cannabinoid, the receptor involved, and the physiological context in which the signal occurs.
Although the term cannabinoid is immediately associated with cannabis, its meaning is broader. There are cannabinoids produced by the body itself, plant-based compounds, and molecules synthesized in the laboratory, all of which are capable of primarily modulating the ECS (and, in some cases, other related signaling pathways).
Types of cannabinoids and compounds that modulate the endocannabinoid system
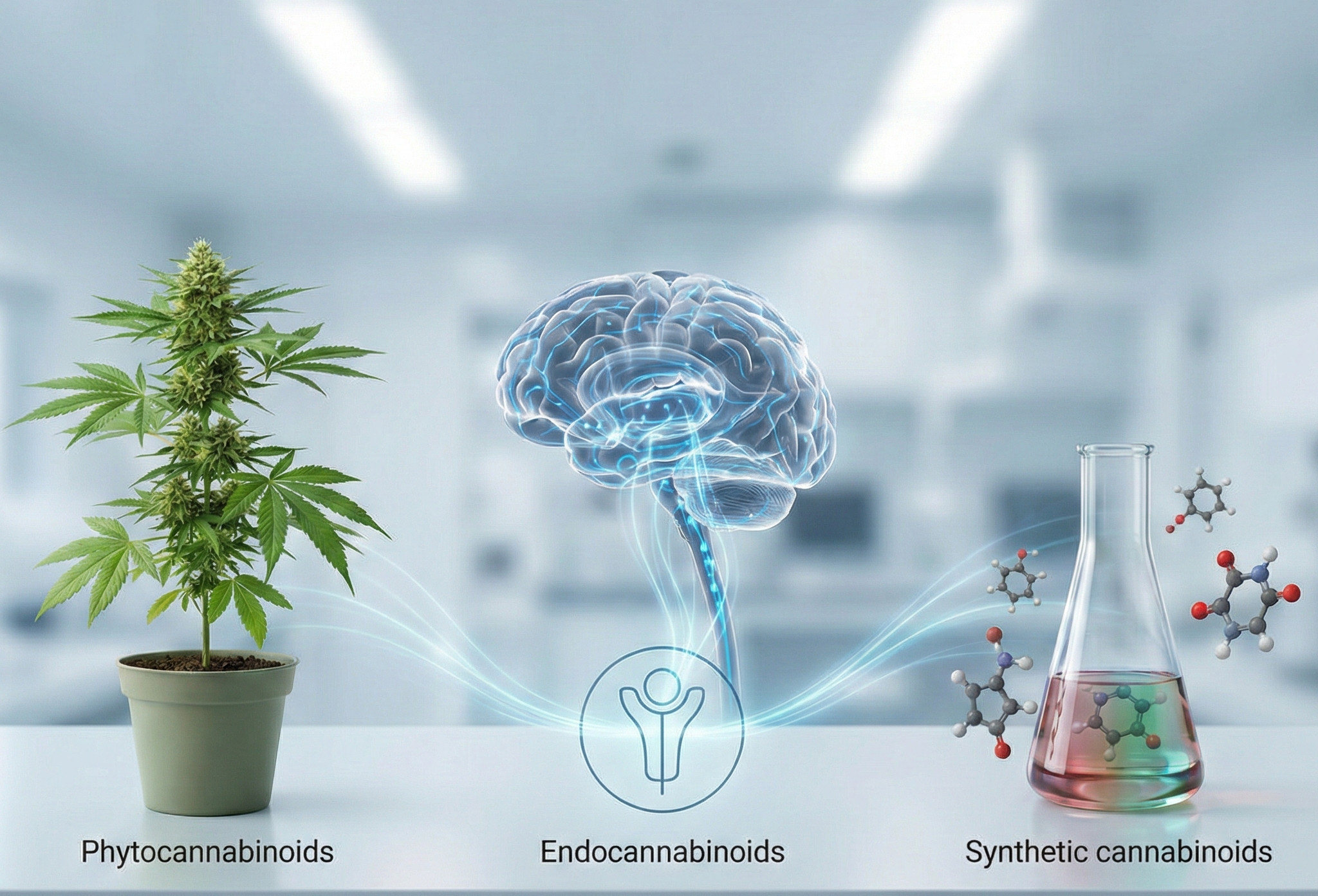 Although they interact with the same receptors, the origin of these compounds varies drastically: from natural biosynthesis in the plant or the brain, to synthetic origin.
Although they interact with the same receptors, the origin of these compounds varies drastically: from natural biosynthesis in the plant or the brain, to synthetic origin.
Endocannabinoids: those produced by the body
Endocannabinoids are cannabinoids synthesized endogenously by the human body and other mammals. They are not stored in large quantities but are produced on demand from lipid precursors present in cell membranes.
The two main characterized endocannabinoids are anandamide (AEA) and 2-arachidonoylglycerol (2-AG). Both actively participate in the modulation of neurotransmission, stress response, appetite, memory, and inflammatory processes. Unlike many classical neurotransmitters, endocannabinoids act retrogradely, regulating the release of other chemical messengers.
The presence of endocannabinoids has been described in breast milk, and it has been proposed that they could participate in early behaviors such as suckling and aspects of development; however, the details of these mechanisms in humans remain an active area of research.
Phytocannabinoids: those of plant origin
Phytocannabinoids are cannabinoids synthesized by plants, with Cannabis sativa L. being one of the richest and most diverse sources known to date. Although compounds with cannabimimetic activity have been identified in other plant species, cannabis stands out for the quantity, variety, and potency of the phytocannabinoids it produces.
For decades it was assumed that these compounds were exclusive to cannabis. While it is now known that this is not strictly true, this plant remains the most pharmacologically and biologically relevant regarding the endocannabinoid system, which is why it constitutes the main focus of analysis in this field.
Synthetic cannabinoids: laboratory-designed molecules
Synthetic cannabinoids are compounds created artificially with the aim of interacting with cannabinoid receptors. Some of them have been developed for therapeutic purposes, such as dronabinol, used in specific clinical contexts under medical supervision.
A distinction must be made between synthetic cannabinoids used as drugs (e.g., dronabinol in specific clinical contexts) and recreational mixtures like K2/Spice. In the latter, the composition is often uncertain, and some compounds can activate CB1 intensely (even as full agonists), which is associated with a profile of adverse effects and toxicity that is more unpredictable than that of THC.
Mimetic cannabinoids or cannabimimetics
There are compounds that, without chemically belonging to the cannabinoid family, mimic their action or modulate the endocannabinoid system. These are found in various plants and foods.
Beta-caryophyllene, present in black pepper, cloves, and cannabis, is one of the most studied examples for its selective affinity for CB2 receptors. Other compounds present in echinacea, cacao, or certain truffles also interact indirectly with this system, reinforcing its role as a biochemical interface between diet, environment, and human physiology.
The Endocannabinoid System (ECS)
The endocannabinoid system (ECS) is a complex regulatory network composed of cannabinoid receptors, endocannabinoids (lipid messengers produced by the body), and enzymes responsible for their synthesis and degradation. Together, it contributes to homeostasis, adjusting multiple physiological processes in the face of internal or external changes.
A distinctive feature of the ECS is that many endocannabinoids are synthesized "on demand" from membrane lipids and act locally for a short time. In the nervous system, they can function as retrograde signals: the postsynaptic neuron releases endocannabinoids that act on presynaptic terminals, modulating neurotransmitter release and synaptic plasticity.
CB1 receptors are expressed predominantly in the central nervous system, especially in regions involved in cognition, memory, motor control, and pain perception. Their activation explains much of the neurological effects of THC, as it modifies synaptic signaling and the balance of neuronal circuits involved in those functions.
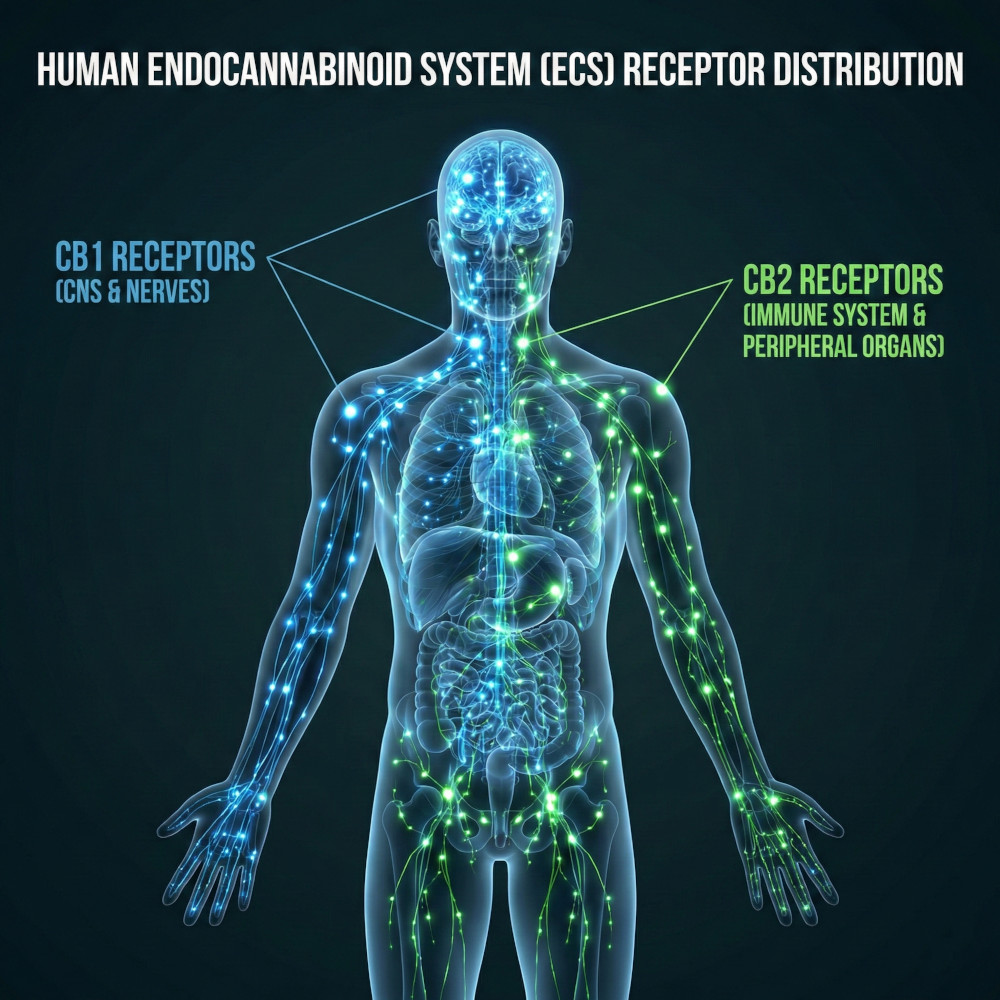 CB1 receptors (blue) predominate in the brain and central nervous system, while CB2 receptors (green) are more dispersed, with greater presence in the immune system and peripheral tissues.
CB1 receptors (blue) predominate in the brain and central nervous system, while CB2 receptors (green) are more dispersed, with greater presence in the immune system and peripheral tissues.
CB2 receptors are located mainly in immune system cells and peripheral tissues. Their activation is associated with the modulation of the immune response and inflammatory and tissue protection processes, without a typical intoxicating profile.
Finally, the enzymes that degrade endocannabinoids—such as FAAH (associated with anandamide) and MAGL (associated with 2-AG)—help regulate the endocannabinoid "tone." This enzymatic component is key to understanding that the ECS depends not only on receptors but also on how long endogenous signals persist in each tissue.
The phytocannabinoids of cannabis
Where and how plant cannabinoids are produced
 The highest concentration of phytocannabinoids (such as THC or CBD) is found inside the spherical heads of the trichomes
The highest concentration of phytocannabinoids (such as THC or CBD) is found inside the spherical heads of the trichomes
Phytocannabinoids are synthesized mainly in glandular trichomes, specialized structures that cover primarily the flowers and nearby leaves. These secretory glands produce a resin rich in cannabinoids, terpenes (responsible for aroma and flavor), and flavonoids, and also act as a chemical interface with the environment (for example, against radiation, desiccation, or herbivory), which helps explain why their abundance and composition vary according to genetics and growing conditions.
From a biosynthetic point of view, most cannabinoids derive from a common precursor: cannabigerolic acid (CBGA). Through specific enzymatic pathways, CBGA is transformed into THCA, CBDA, or CBCA, depending on the plant's genetic expression; this is why we speak of chemotypes (plants with THC dominance, CBD dominance, or mixed profiles), which are more informative than traditional commercial labels when seeking to predict the effect.
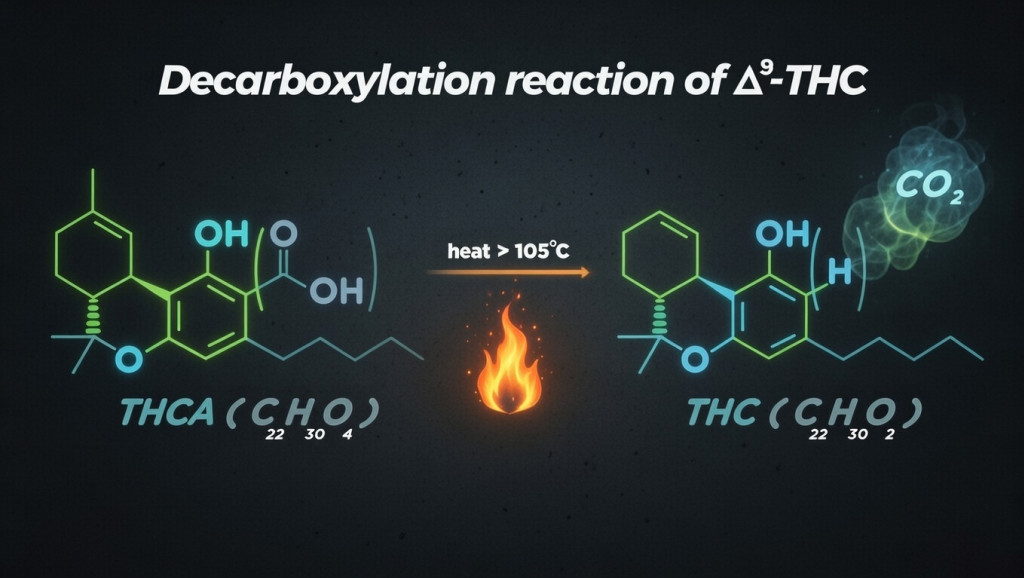
In the fresh plant, acidic forms such as THCA or CBDA predominate, which do not produce the intoxicating profile characteristic of THC. The application of heat triggers decarboxylation, transforming those acids into their neutral forms (e.g., THCA→THC and CBDA→CBD), decisively changing their pharmacological effect. This detail is key to interpreting differences between raw flower, unactivated extractions, and products subjected to heat: the "potential" of a material depends not only on the total percentage but on how much is already in neutral form and how it is prepared and consumed.
If you want to understand why heat transforms THCA into THC and how it affects the final effect, you will be interested in this article:
Decarboxylation transforms acidic cannabinoids (THCA, CBDA) into their active forms through heat. Temperatures of 110-115°C for 40-45 minutes optimize conversion while preserving terpenes and minim...
The major cannabinoids of marijuana
THC (delta-9-tetrahydrocannabinol) is the main intoxicating phytocannabinoid of cannabis. Its action as a partial agonist of CB1 receptors, highly expressed in the central nervous system, explains much of its effects: changes in sensory and time perception, mood modulation, increased appetite, and transient alterations in functions such as attention, working memory, and motor coordination. The intensity and profile of the effect depend on variables such as dose, route of administration (inhaled or oral), tolerance, and the presence of other cannabis compounds that may modulate the response.
CBD (cannabidiol) presents a distinct pharmacological profile. It is not intoxicating and does not act as a classical CB1 agonist; its influence on the endocannabinoid system is more indirect and "regulatory," modulating the system's tone through different targets (including enzymes and non-exclusively cannabinoid receptors). In practical terms, this helps understand why CBD does not produce the "high" typical of THC and why, in some people and formulations, it can soften certain undesirable effects of THC (such as nervousness or a feeling of lack of control), although that result is not automatic and depends on actual proportions and doses.
Minor cannabinoids and their emerging relevance
CBG (cannabigerol) occupies a special position because, in its acidic form (CBGA), it is the biosynthetic starting point from which the plant generates other families (THCA, CBDA, CBCA). In pharmacological terms, CBG is not intoxicating in the classical sense of THC and is studied for its possible role as a modulator in processes such as inflammation and pain, in addition to interest in its antibacterial and neuroprotective activities described in preclinical research. Its relationship with ocular parameters has also been explored, although it is best formulated as a field under investigation rather than an established effect in humans.
CBN (cannabinol) is not usually a "dominant" cannabinoid in the fresh plant: it appears primarily when THC degrades through aging and oxidation, which is why its presence may increase in poorly preserved or heavily cured material. It is frequently associated with a more sedative profile, but this idea requires nuance: in real products, the sensation of drowsiness may depend as much on the context (total dose, tolerance, time of day) as on the rest of the extract (other cannabinoids and terpenes).
CBN is not a cannabinoid that the plant produces: it forms when THC oxidises. For decades that made it synonymous with degraded cannabis. It is now understood as something different: a sedative pr...
THCV (tetrahydrocannabivarin) is one of the most interesting "varinic" cannabinoids due to its dose-dependent pharmacology. At low doses, it can behave as a functional antagonist or modulator of CB1 in certain models, while at higher doses, it may show effects closer to cannabinoid activation. In layman's terms, this translates to research into its possible relationship with appetite and metabolism, and subjectively, some people describe effects that are "clearer" or less sedative than those of THC, although these experiences are not uniform and depend on the chemotype and formulation.
CBC (cannabichromene) is a non-intoxicating cannabinoid that usually appears in lower concentrations but has gained interest for its possible role in modulating pain and inflammation. Its action seems to involve, at least in part, targets other than CB1/CB2 (for example, channels involved in sensory signaling), making it a candidate for "modulator" within broad-spectrum formulations. On an applied level, CBC and THCV (along with CBG and other minors) are driving a stage of genetic selection oriented toward more specific chemical profiles, beyond the THC/CBD binomial.
 Summary of the most relevant chemical families (such as THC, CBD, and their acidic forms) and the therapeutic properties attributed to them in current research.
Summary of the most relevant chemical families (such as THC, CBD, and their acidic forms) and the therapeutic properties attributed to them in current research.
The Entourage Effect
The so-called entourage effect is a pharmacological hypothesis: it posits that the effects of cannabis do not depend solely on THC or CBD separately, but on the ensemble of compounds present in a preparation (phytocannabinoids, terpenes, and other minor components). This idea is used to explain why two products with a similar THC percentage can generate distinct experiences, and why some full-spectrum extracts are perceived as more "rounded" than isolates.
Even so, language should be nuanced: it is not always a guaranteed "synergy." In practice, interactions between compounds can be synergistic, additive, or even antagonistic, and their expression depends on variables such as dose, relative proportions, route of administration, tolerance, and individual differences.
From a useful point of view for the reader, the entourage effect invites looking beyond a single number (for example, "THC %"). In the therapeutic realm and in the development of varieties or extracts, it suggests that the reproducibility of the complete chemical profile matters as much as potency, but without turning it into a commercial argument: verifiable composition, stability, and consistent dosing remain the basis for speaking with rigor.
In the following article, we explain the entourage effect in depth:
Discover what the entourage effect of cannabis is: how cannabinoids, terpenes and flavonoids work together to enhance therapeutic effects.
Cannabinoids are part of a signaling system deeply integrated into human physiology. The endocannabinoid system acts as a fundamental regulatory axis connecting the nervous system, the immune system, and metabolism.
Cannabis is relevant because it produces phytocannabinoids capable of effectively interacting with this system, not because it created it. Understanding this interaction from a scientific perspective allows for approaching the study of cannabis and its compounds with rigor, moving away from simplistic approaches.
Sources and references
- Lu, H.-C., & Mackie, K. (2021). Review of the Endocannabinoid System. Biological Psychiatry: Cognitive Neuroscience and Neuroimaging, 6(6), 607–615. https://doi.org/10.1016/j.bpsc.2020.07.016 (PMC)[1][2]
- De Petrocellis, L., Cascio, M. G., & Di Marzo, V. (2004). The endocannabinoid system: a general view and latest additions. British Journal of Pharmacology, 141(5), 765–774. https://doi.org/10.1038/sj.bjp.0705666 (PMC)[3][4]
- Di Marzo, V., Bifulco, M., & De Petrocellis, L. (2004). The endocannabinoid system and its therapeutic exploitation. Nature Reviews Drug Discovery, 3(9), 771–784. <https://doi.org/10.1038/nrd1495>[5]
- Devane, W. A., Hanus, L., Breuer, A., et al. (1992). Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science. (Classic article: anandamide/AEA). PubMed: 1470919[6]
- Russo, E. B. (2011). Taming THC: potential cannabis synergy and phytocannabinoid-terpenoid entourage effects. British Journal of Pharmacology, 163(7), 1344–1364. https://doi.org/10.1111/j.1476-5381.2011.01238.x (PMC)[7][8]
- Gertsch, J., Leonti, M., Raduner, S., et al. (2008). Beta-caryophyllene is a dietary cannabinoid. Proceedings of the National Academy of Sciences of the United States of America (PNAS). <https://doi.org/10.1073/pnas.0803601105>[9][10]
- Long, J. Z., Li, W., Booker, L., et al. (2009). Dual blockade of FAAH and MAGL identifies behavioral processes regulated by endocannabinoid crosstalk in vivo. PNAS. <https://doi.org/10.1073/pnas.0909411106>[11]
- Targeting Endocannabinoid Signaling: FAAH and MAG Lipase… (2021). Annual Review of Pharmacology and Toxicology. <https://doi.org/10.1146/annurev-pharmtox-030220-112741>[12]
- Wang, M., Wang, Y.-H., Avula, B., et al. (2016). Decarboxylation Study of Acidic Cannabinoids: A Novel Approach Using Ultra-High-Performance Supercritical Fluid Chromatography/Photodiode Array-Mass Spectrometry. Cannabis and Cannabinoid Research, 1(1), 262–271. https://doi.org/10.1089/can.2016.0020 (PMC)[13][14]
- Castaneto, M. S., Gorelick, D. A., Desrosiers, N. A., et al. (2014). Synthetic Cannabinoids: Pharmacology, Behavioral Effects, and Abuse Potential. (Review; useful for differentiating THC vs high-efficacy synthetics). https://doi.org/10.1097/FBP.0000000000000034 (PMC)[15]