When cannabis is cultivated or acquired, the buds mainly contain cannabinoids in their acidic form: THCA (tetrahydrocannabinolic acid) and CBDA (cannabidiolic acid), among others. These precursor compounds are not psychoactive in the case of THCA, nor do they have the same bioavailability as their neutral forms. Decarboxylation is the chemical process that transforms these acidic cannabinoids into their active forms: THC, CBD, and other cannabinoids known for their therapeutic and recreational effects.
Decarboxylation is the chemical process that transforms these acidic cannabinoids into their active forms
What is decarboxylation?
Decarboxylation is a chemical reaction in which a carboxyl group (COOH) is removed from a molecule in the form of carbon dioxide (CO₂). In the context of cannabis, this reaction converts acidic cannabinoids into their neutral analogues. For example, THCA (C₂₂H₃₀O₄) loses a carboxyl group to become THC (C₂₁H₃₀O₂) plus CO₂. This transformation not only changes the chemical structure but also radically alters the pharmacological properties of the molecule.
THCA, predominant in fresh cannabis, has a molecular weight of approximately 358 g/mol, while the resulting THC weighs around 314 g/mol. This difference in mass represents precisely the lost carboxyl group. Although THCA possesses some therapeutic properties of its own, such as anti-inflammatory and neuroprotective effects documented in preliminary research, it lacks the psychoactive capacity of THC due to its poor affinity for the CB1 receptors of the endocannabinoid system.
The kinetics of the decarboxylation chemical reaction
Decarboxylation is a temperature-dependent reaction that occurs naturally but extremely slowly at room temperature. The living cannabis plant produces mainly acidic cannabinoids, and only small amounts spontaneously convert to their neutral forms during drying and curing. For the conversion to be significant and controlled, the application of heat is required.
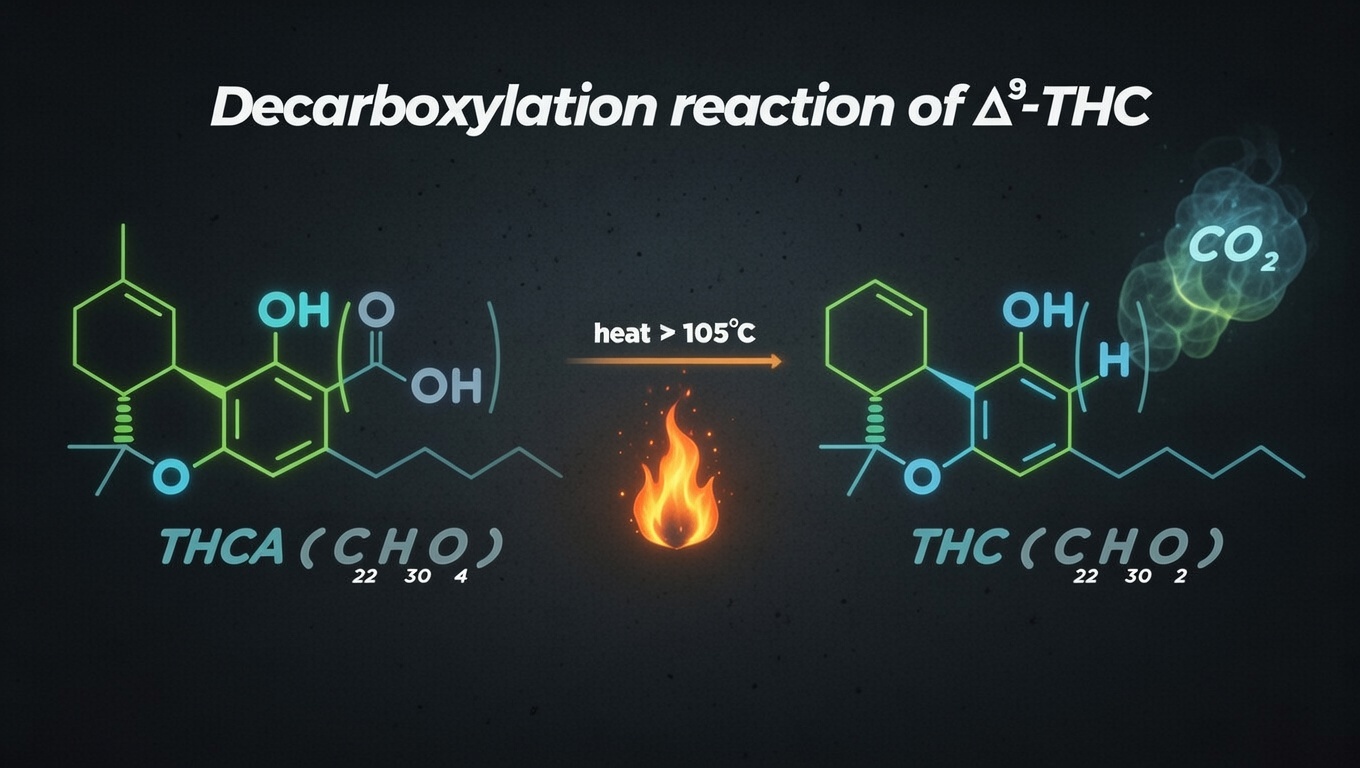
Scientific research has identified optimal temperature ranges to maximize decarboxylation. A study published in the Journal of Chromatography A by Wang and colleagues in 2016 demonstrated that THCA decarboxylation reaches maximum efficiency between 105°C and 120°C. At 110°C, approximately 70% of THCA converts to THC after 30 minutes, while at temperatures above 140°C, although the reaction is faster, degradation of THC into cannabinol (CBN) begins, a cannabinoid with different properties and generally considered less desirable.
At temperatures above 140°C, degradation of THC into cannabinol (CBN) begins
Exposure time is equally critical. At 100°C, complete decarboxylation may require up to 60 minutes, while at 120°C the process can be completed in approximately 30-40 minutes. However, maintaining excessive temperatures even for short periods can degrade not only cannabinoids but also terpenes, volatile aromatic compounds that contribute to the entourage effect and the organoleptic properties of cannabis.
Cannabis decarboxylation methods
The most common form of decarboxylation is that which occurs during smoking or vaporization of cannabis. When combusted, the temperature at the lit end of a cigarette can exceed 800°C, causing instantaneous decarboxylation but also the destruction of many compounds and the generation of potentially harmful combustion byproducts. Vaporization, which operates between 160°C and 220°C depending on the device, allows more controlled decarboxylation with less production of degradation compounds.
| Acid | Cannabinoid | Temperature (°C) | Temperature (°F) | Oven time (min) |
|---|---|---|---|---|
| THCa | THC | 95 | 200 | 50 |
| THCa | THC | 110 | 230 | 25 |
| THCa | THC | 130 | 265 | 7 |
| CBDa | CBD | 110 | 230 | 40 |
| CBDa | CBD | 130 | 265 | 12 |
| CBGa | CBG | 110 | 230 | 40 |
For the production of extracts, tinctures, edibles, or oils, prior decarboxylation of plant material is required. The most widespread method consists of baking ground cannabis in a conventional oven. Scientific literature suggests that distributing plant material in a thin layer on a tray and baking it at 115°C for 40-45 minutes produces optimal results in terms of THCA to THC conversion while preserving the integrity of other compounds.
Researchers at Leiden University, in a study published in Phytochemistry in 2017, examined decarboxylation using different methods and found that the use of a water bath or sous-vide (vacuum cooking) at controlled temperatures of 100-105°C for 60-90 minutes can offer advantages in terms of homogeneity and terpene preservation, although it requires longer times.
Variables affecting decarboxylation
Several factors influence the efficiency of decarboxylation beyond temperature and time. The moisture content of plant material plays an important role: cannabis with high water content requires longer exposure time because part of the thermal energy is used to evaporate water. For this reason, the material should be adequately dried before decarboxylation, although excessive drying can make the material too brittle and affect terpene conservation.
Limonene is a monoterpene present in cannabis with anti-inflammatory, antioxidant and anxiolytic properties. It increases serotonin and dopamine, participates in the entourage effect with cannabino...
The degree of grinding also matters. Fine grinding increases the surface area exposed to heat, facilitating more uniform decarboxylation, but can accelerate degradation if temperature is not carefully controlled. The presence of oxygen is another relevant factor: decarboxylation in an oxygen-rich atmosphere can promote unwanted oxidation reactions that degrade cannabinoids. Some commercial methods employ inert or vacuum atmospheres to minimize this effect.
Beyond THC: decarboxylation of other cannabinoids
Although the focus is usually on the conversion of THCA to THC, other cannabinoids undergo similar processes. CBDA decarboxylates into CBD with comparable kinetics, though slightly more heat-resistant. This means that CBD requires similar or slightly higher temperatures and comparable times for complete conversion. CBGA (cannabigerolic acid), the precursor of many cannabinoids, also decarboxylates into CBG (cannabigerol) following similar patterns.
A less well-known aspect is that partial decarboxylation may be desirable in certain contexts. Some research suggests that the combination of acidic and neutral cannabinoids could offer synergistic effects. THCA, for example, has shown in preclinical studies antiemetic, neuroprotective, and anti-inflammatory properties independent of THC, which has led some medicinal extract producers to seek profiles with controlled mixtures of both forms.
Implications for dosing
Understanding decarboxylation is crucial for precise dosing, especially in medicinal applications. One gram of cannabis with 20% THCA does not equal 200 mg of available THC without decarboxylation. Theoretically, complete conversion of THCA to THC results in approximately 87.7% of the original weight due to the loss of the carboxyl group. Thus, 200 mg of THCA would produce approximately 175 mg of THC under ideal conditions. In practice, due to losses from degradation and conversion efficiency, yields are typically 70-85% in home processes.
Practical example: 5 g of cannabis at 20% THCA contains 1000 mg of THCA. After decarboxylation, the theoretical maximum is ~877 mg of THC, but in home processes the usual result is 700–800 mg effective.
This variability underscores the importance of standardization in medicinal products. Modern cannabinoid analysis laboratories quantify both acidic and neutral forms, and some report "total THC" calculated assuming complete decarboxylation to give an estimate of the maximum psychoactive potential of the material.
Safety and storage considerations
Decarboxylation generates vapor that contains terpenes and potentially small amounts of volatilized cannabinoids. It is recommended to perform the process in well-ventilated spaces. The characteristic smell of cannabis intensifies during decarboxylation due to the release of terpenes, which can be a consideration in environments where discretion is important.
Once decarboxylated, cannabis and its derivatives are more susceptible to degradation. THC is particularly sensitive to light, oxygen, and heat. Prolonged exposure to UV light can convert THC into CBN, altering the effects profile. Therefore, decarboxylated products should be stored in opaque, airtight containers in cool, dark places to maximize their shelf life.
Sources and references
- Wang, M., Wang, Y. H., Avula, B., Radwan, M. M., Wanas, A. S., van Antwerp, J., ... & Khan, I. A. (2016). Decarboxylation study of acidic cannabinoids: a novel approach using ultra-high-performance supercritical fluid chromatography/photodiode array-mass spectrometry. Cannabis and Cannabinoid Research, 1(1), 262-271.
- Dussy, F. E., Hamberg, C., Luginbühl, M., Schwerzmann, T., & Briellmann, T. A. (2005). Isolation of Δ9-THCA-A from hemp and analytical aspects concerning the determination of Δ9-THC in cannabis products. Forensic Science International, 149(1), 3-10.
- Veress, T., Szanto, J. I., & Leisztner, L. (1990). Determination of cannabinoid acids by high-performance liquid chromatography of their neutral derivatives formed by thermal decarboxylation. Journal of Chromatography A, 520, 339-347.
- Perrotin-Brunel, H., Pérez, P. C., van Roosmalen, M. J., van Spronsen, J., Witkamp, G. J., & Peters, C. J. (2011). Decarboxylation of Δ9-tetrahydrocannabinol: kinetics and molecular modeling. Journal of Molecular Structure, 987(1-3), 67-73.
- Hanuš, L. O., Meyer, S. M., Muñoz, E., Taglialatela-Scafati, O., & Appendino, G. (2016). Phytocannabinoids: a unified critical inventory. Natural Product Reports, 33(12), 1357-1392.
- Grotenhermen, F. (2003). Pharmacokinetics and pharmacodynamics of cannabinoids. Clinical Pharmacokinetics, 42(4), 327-360.
- Palmieri, B., Laurino, C., & Vadalà, M. (2019). Short-term efficacy of CBD-enriched hemp oil in girls with dysautonomic syndrome after human papillomavirus vaccination. Isr Med Assoc J, 21(2), 125-130.